When you explore the best laboratory chemicals available today, you’ll discover that these essential compounds form the backbone of scientific research, quality control, and industrial applications across countless industries. Laboratory chemicals with superior properties and proven uses across industries enable researchers and professionals to achieve breakthrough results in pharmaceuticals, biotechnology, environmental testing, and manufacturing sectors.
Whether you’re conducting analytical testing, developing new products, or performing routine laboratory procedures, selecting quality lab-grade chemicals is crucial for achieving accurate and reliable results. At Oozechem.com, we understand that the properties and uses of laboratory chemicals directly impact the success of your research and industrial applications.
Understanding Laboratory Chemicals Classifications
Laboratory chemicals can be broadly categorized based on their intended use and purity levels. Analytical grade chemicals represent the highest purity standard, typically containing 99.5% or higher active ingredient concentration. These chemicals undergo rigorous testing and certification processes to ensure minimal impurities that could interfere with sensitive analytical procedures.
Technical grade chemicals offer a cost-effective solution for general laboratory work where ultra-high purity isn’t critical. These chemicals maintain good quality standards while being more economical for routine applications such as cleaning, preparation work, and non-critical synthetic procedures.
Reagent grade chemicals strike a balance between purity and practicality, making them ideal for most synthetic chemistry applications, educational laboratories, and quality control testing where consistent results are essential but maximum purity isn’t required. When you explore the best laboratory chemicals in this category, you’ll find they offer reliable performance for diverse uses across industries while maintaining cost-effectiveness.

Essential Properties to Consider
When selecting laboratory chemicals, several key properties determine their suitability for specific applications and uses across industries. Understanding these properties helps you discover quality lab-grade chemicals that will deliver optimal performance in your specific applications. Chemical purity remains the most critical factor, as even trace impurities can significantly impact experimental results, particularly in analytical chemistry and pharmaceutical research.
Stability and shelf life directly affect the long-term reliability of your chemical inventory. Some chemicals degrade rapidly when exposed to light, moisture, or air, requiring special storage conditions and frequent replacement. Understanding these stability characteristics helps laboratories maintain chemical integrity and avoid experimental failures.
Solubility characteristics play a vital role in determining how chemicals can be used in different solvents and reaction conditions. Water-soluble chemicals offer versatility for aqueous systems, while organic-soluble compounds are essential for non-polar reaction environments.
Physical properties such as melting point, boiling point, and density provide crucial information for handling, storage, and application procedures. These properties also serve as quality indicators, helping verify chemical identity and purity upon receipt.
Advanced Laboratory Applications Table
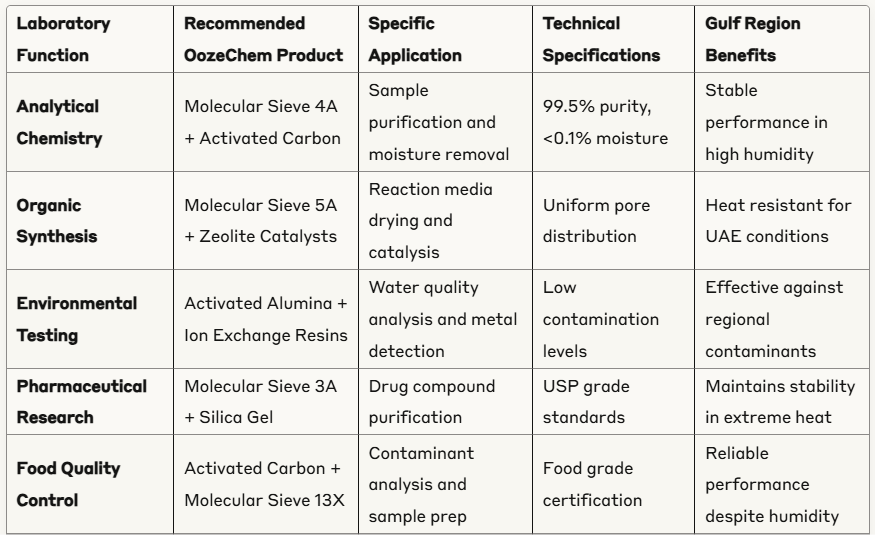
Common Laboratory Chemical Categories and Their Uses
Acids and Bases
Acids and bases form fundamental chemical groups used across virtually all laboratory applications. These essential reagents serve multiple purposes including digestion procedures, pH adjustment, and synthetic chemistry applications.
Common Acids:
- Hydrochloric acid – Used for pH adjustment, metal digestion, and analytical procedures
- Sulfuric acid – Essential for organic synthesis, dehydration reactions, and battery acid applications
- Nitric acid – Utilized in metal etching, oxidation reactions, and explosive manufacturing
- Acetic acid – Applied in buffer preparations, organic synthesis, and preservation
Common Bases:
- Sodium hydroxide – Used for neutralization reactions, soap manufacturing, and pH adjustment
- Potassium hydroxide – Essential for biodiesel production, battery electrolytes, and chemical synthesis
- Ammonia solutions – Applied in cleaning products, fertilizer production, and refrigeration systems
- Calcium hydroxide – Utilized in water treatment, construction materials, and food processing

Organic Solvents
Organic solvents enable dissolution, extraction, and purification of countless compounds. Each solvent offers unique solubility properties and compatibility with different chemical systems.
Polar Solvents:
- Methanol – Excellent for polar compound extraction and HPLC mobile phases
- Ethanol – Used in pharmaceutical formulations, extractions, and as a green solvent alternative
- Acetone – Applied in paint thinning, degreasing, and polymer dissolution
- Isopropanol – Essential for cleaning electronics, disinfection, and cosmetic formulations
Non-Polar Solvents:
- Dichloromethane – Used for organic extractions, paint stripping, and pharmaceutical synthesis
- Hexane – Applied in oil extraction, chromatography, and polymer processing
- Toluene – Utilized in paint production, adhesives, and chemical synthesis
- Chloroform – Used in pharmaceutical extraction and as an industrial solvent
Buffer Systems
Buffer systems maintain stable pH conditions essential for biological assays, enzyme reactions, and analytical procedures. These systems provide reliable pH control across different ranges.
Phosphate Buffers:
- Monosodium phosphate – Used for pH range 1.1-3.1 in biological applications
- Disodium phosphate – Applied for pH range 6.1-8.1 in cell culture media
- Trisodium phosphate – Utilized for pH range 11.1-12.1 in alkaline conditions
Biological Buffers:
- Tris buffer – Essential for pH range 7.0-9.0 in biochemical research
- HEPES buffer – Used for pH range 6.8-8.2 in cell culture applications
- MES buffer – Applied for pH range 5.5-6.7 in protein studies
Indicators and Dyes
Indicators and dyes enable visual detection of chemical changes, endpoint determination in titrations, and qualitative analysis procedures.
pH Indicators:
- Phenolphthalein – Changes from colorless to pink at pH 8.2-10.0
- Methyl orange – Transitions from red to yellow at pH 3.1-4.4
- Bromothymol blue – Shifts from yellow to blue at pH 6.0-7.6
- Universal indicator – Provides color changes across the entire pH range
Redox Indicators:
- Methylene blue – Used in redox titrations and biological staining
- Potassium permanganate – Applied as both oxidizing agent and indicator
- Iodine solution – Essential for starch detection and redox reactions
Quality Standards and Certification
Reputable chemical suppliers maintain strict quality control standards backed by comprehensive testing protocols. Certificate of Analysis (COA) documentation accompanies each chemical batch, providing detailed information about purity levels, impurity profiles, and physical property verification.
ISO certification indicates that suppliers follow internationally recognized quality management systems, ensuring consistent product quality and reliable supply chains. This certification becomes particularly important for laboratories requiring traceability and documentation for regulatory compliance.
GMP compliance (Good Manufacturing Practices) ensures that chemicals intended for pharmaceutical or food-related applications meet stringent production standards. These chemicals undergo additional testing and documentation requirements to support their use in regulated industries.
Safety Considerations and Handling
Laboratory chemical safety extends beyond basic protective equipment to encompass proper storage, handling procedures, and emergency response protocols. Safety Data Sheets (SDS) provide comprehensive information about chemical hazards, first aid procedures, and disposal requirements.
Storage compatibility requires careful consideration of chemical interactions that could lead to dangerous reactions. Acids should be stored separately from bases, oxidizers must be isolated from organic materials, and water-reactive chemicals require moisture-free environments.
Personal protective equipment selection depends on specific chemical properties and exposure risks. While basic lab coats, safety glasses, and gloves provide general protection, some chemicals require specialized equipment such as fume hoods, respirators, or chemical-resistant clothing.
Choosing the Right Chemical Supplier
Selecting a reliable chemical supplier significantly impacts laboratory efficiency, safety, and results quality. Technical support capabilities help laboratories troubleshoot applications, optimize procedures, and select appropriate chemical grades for specific requirements.
Supply chain reliability ensures consistent chemical availability, preventing research delays and maintaining laboratory productivity. Established suppliers with robust inventory management and logistics capabilities provide greater confidence in meeting ongoing chemical needs.
Regulatory compliance support becomes increasingly important as laboratories face evolving safety and environmental regulations. Suppliers who understand regulatory requirements can provide guidance on proper documentation, handling procedures, and disposal methods.
Conclusion
Successful laboratory operations depend on selecting high-quality chemicals that match specific application requirements while maintaining safety and cost-effectiveness. By understanding chemical properties, quality standards, and supplier capabilities, laboratories can build reliable chemical inventories that support accurate, reproducible results.
At Oozechem.com, we understand that every laboratory has unique requirements, whether you’re conducting cutting-edge research, performing routine quality control, or supporting educational programs. When you explore the best laboratory chemicals in our comprehensive catalog, you’ll discover quality lab-grade chemicals that meet rigorous standards for properties and uses across industries. Our commitment to providing superior laboratory chemicals, combined with expert technical support, ensures that you receive the right chemicals for your applications, backed by the reliability and service excellence your laboratory deserves.
